
Luxmanan Selvanesan
Contact:
HortResearch Mt Albert,
120 Mt Albert Road,
Auckland
Department of Biochemistry,
University of Otago,
Dunedin
Email: lux10selva@yahoo.com
Supervisors:
Dr. Elspeth MacRae, HortResearch, EMacRae@hortresearch.co.nz
Dr. Julian-Eaton-Rye, University of Otago, jjer@sanger.otago.ac.nz
My PhD project is designed to identify the function of the different isozymes of sucrose phosphate synthase (SPS) found in A. thaliana and to compare these isozymes with cyanobacterial forms. As part of the project we will carry-out comprehensive evolutionary analysis between different families of SPS. The project ultimately aims to characterise the SPS isoenzymes and understand why they have evolved. The project is funded through Marsden grant which includes a collaboration with overseas laboratories.
SPS converts UDP-glucose and fructose 6-phosphate to sucrose phosphate. Sucrose phosphate is then converted to sucrose by sucrose phosphate phosphatase. Sucrose is then available for transport to other plant tissues or for transfer to organelles such as vacuoles. In addition to its role as the transportable form of carbon in plants, sucrose also functions in signal transduction and plays a critical role in protection against stress-induced damage by low temperature or osmotic alteration.
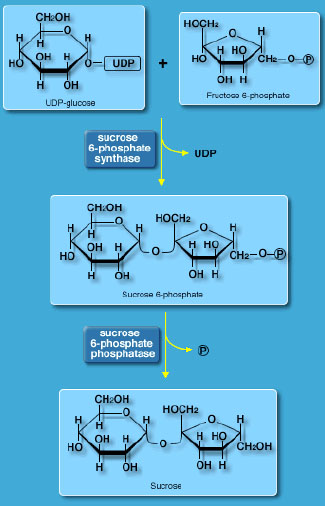
It has been shown that these enzymes are restricted to cyanobacterial species (the plastid progenitor) and plants, and that they are multidomain proteins with a modular structure. Furthermore, the MacRae lab at HortResearch has also been shown that higher plant SPS belongs to three different families based on protein sequence.
They have also shown that subfamilies can also occur. This will be verified as part of this project though phylogenetic analysis using all the publicly available SPS protein sequences and other genes that will be isolated from lower plants by collaborators.
Function of these different isoenzymes in higher plants is not well understood. Apart from studying such an enzyme with evolutionary significance is a merit in itself, the importance of sucrose in higher plants makes it an attractive enzyme to research.
|